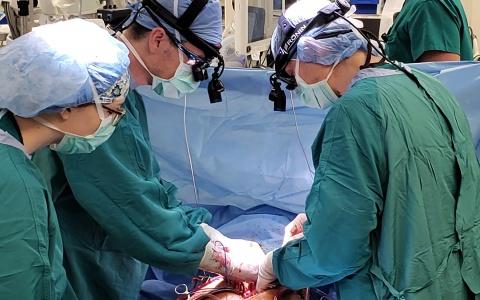
Photo: Drs. Jacob Schroder and Carmelo Milano transplant the first donation after circulatory death (DCD) heart in the United States
Approximately 6 million patients in the United States live with heart failure. Of those, 10% will progress to end-stage heart failure. While there is no cure, heart transplantation can bring hope to patients when other treatments no longer can. But due to the nationwide organ shortage, most patients do not receive life-saving transplants fast enough. In 2019, only 3,551 heart transplants were performed, with patients waiting up to a year or more for a transplant.
On December 1, 2019, Drs. Jacob Schroder, Benjamin Bryner, and Carmelo Milano, Division of Cardiovascular and Thoracic Surgery, performed the first heart transplant in the United States using a heart from an adult donor after circulatory death (DCD). This type of organ donation has been well established for kidney, liver, pancreas, and lung transplants, but hearts from DCD donors have not been used for transplant until now. With the introduction of DCD hearts to the donor pool, more patients could receive heart transplants faster.
“There’s a whole group in the middle,” says Dr. Jacob Schroder, Director of the Duke Heart Transplantation Program. “The status 4’s especially, patients who have left ventricular assist devices who have been on the list for years. I think that’s how we will transplant those patients.”

Donating After Circulatory Death
Heart transplants are typically performed using hearts from organ donors after brain death when neurological function is lost following a devastating injury, such as a trauma, stroke, or drug overdose. DCD organ donors experience the same injuries that leave them on life support with no chance of recovery, but they do not meet the strict criteria for brain death. After the patient’s family agrees to honor their wishes to donate their organs, the patient is taken off life support.
While the United States does not yet have long-term data on DCD heart transplants, institutions in the United Kingdom and Australia have performed this type of transplant for years. In April 2019, Royal Papworth Hospital in Cambridgeshire, United Kingdom, published the results of their first 50 patients following DCD transplant. They compared DCD heart transplants with donation after brain death (DBD) heart transplants and found a 100% 30-day survival rate in both groups with other similar outcomes. According to Dr. Schroder, their wait list time and the size plummeted.
“Of all the DCD donors in the last five years, if you segregate older donors and donors who have known heart conditions, we still think that the donor pool should expand by approximately 30% if we use all those donors,” says Dr. Schroder. “That’s a big deal for us. This will reduce wait list time and wait list mortality with what we hope to be exactly the same survival and clinical results.”
Bringing the Heart Back to Life
The DCD heart transplant was performed at Duke as part of the 2-year EXPAND trial to test a new device that keeps the heart alive outside the body. The TransMedics® Organ Care System™ (OCS™) perfuses the heart with warm, oxygenated blood, simulating conditions in the body. The “heart in a box” device offers a novel method of preserving hearts for transport that reduces ischemia time when the organ lacks oxygen and blood flow. This window of time is critical because patient outcomes worsen when hearts exceed 30 minutes of ischemia because of the onset of heart dysfunction.
“There’s 25 minutes of ischemic time when the heart is not being perfused and then, no matter how far you go with the OCS, it’s being perfused the whole time,” says Dr. Schroder. “To put that into context, if we get a donor at WakeMed, unless we use the helicopter, it takes 30 minutes to drive, and 45-60 minutes to implant the heart. If we go to Puerto Rico using the OCS, the actual ischemic time is shorter than WakeMed. That is the biggest benefit is decreasing that ischemic time.”
In addition to resuscitating the heart, the device allows the surgeons to assess its functional quality. Duke researchers are now looking at ways to improve heart function using novel perfusion methods, including gene therapy and perfusate additives, to rehabilitate the heart before transplantation.
Duke is one of five centers in the United States currently performing DCD heart transplants as part of the trial, which will compare DCD hearts with DBD hearts in terms of patient and graft survival. Following the results of the trial, expected in 2021, TransMedics will seek Food and Drug Administration approval to allow for the wider application of DCD heart transplants in the United States.
“A Gift in a Time of Tragedy”
Donating an organ at the end of life is a selfless act to save the life of a stranger. According to the Organ Procurement and Transplantation Network, one organ donor can save eight lives. This is a difficult decision often made during the most painful moment for a patient and their loved ones. Individuals who wish to donate their organs can communicate their wishes to their loved ones early on to ease the burden.
“This is only possible out of the generosity of donors and their families,” says Dr. Schroder. “This is a gift of life in a time of tragedy. We’re using them because we think these are good hearts to transplant. Our job is to transplant our patients with a good heart as soon as possible, and that’s our goal.”
While the team aims to perform 53 DCD transplants by 2021, Dr. Schroder says they will perform “as many as possible.”
Learn More About Transplant Innovation at Duke Surgery
“Out of Body Experience: Rejuvenating Organs Using Ex Vivo Perfusion”
“Nature vs. Nurture: Transforming Medicine with Gene Therapy”