
Affiliate of the Duke Regeneration Center
Email: david.a.brown@duke.edu
Overview
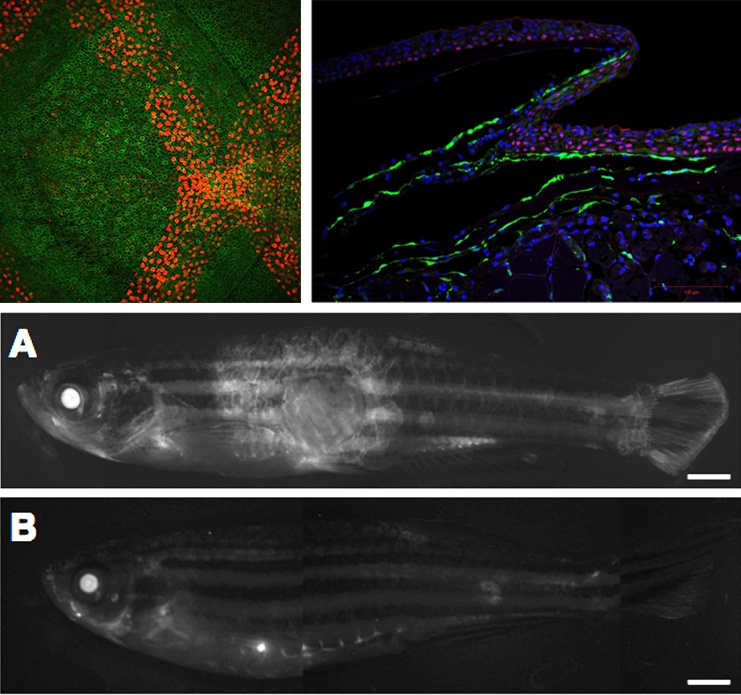
Defects of the skin remain a major clinical problem with few nonsurgical therapies. Skin loss is frequently sustained from trauma, infections, cancer operations, and in chronic wounds. While adult mammalian skin is incapable of full thickness skin regeneration, organisms with innate regenerative capacity such as zebrafish maintain this ability throughout life. Our research program seeks to understand the mechanisms of skin regeneration in zebrafish in order to develop novel regenerative strategies for human wound healing and diseases of the skin.
We use live imaging, transgenic animals, and genomics to study wound healing at the single-cell level. We have a particular focus on basal epidermal keratinocytes, which serve as the stem cell population during epidermal regeneration. Based on prior work that demonstrates the existence of tissue regeneration enhancer elements (TREEs) involved in zebrafish heart and appendage regeneration, the central hypothesis of this project is that re-epithelialization of skin wounds is governed by regeneration-linked regulatory elements that orchestrate coordinated basal keratinocyte behaviors and gene expression.
Program Highlights
- Live confocal imaging of skin wound healing and quantitative tracking
- Chromatin and transcriptome profiling of regenerating skin
Members
- David A. Brown, MD, PhD
- Ben Cox, Department of Cell Biology
- Ken Poss, PhD, Department of Cell Biology
Ongoing Collaborations
- Stefano di Talia, PhD, Department of Cell Biology
- Terry Lechler, PhD, Department of Cell Biology
- Ben Alman, MD, Chair, Department of Orthopaedic Surgery
Collaborate with Us
Contact
David Brown, MD, PhD
Assistant Professor of Surgery
Division of Plastic, Maxillofacial, and Oral Surgery
Department of Surgery
Email: david.a.brown@duke.edu
Publications and Funded Projects
View Dr. Brown's profile to see his publications and funded projects.