Overview
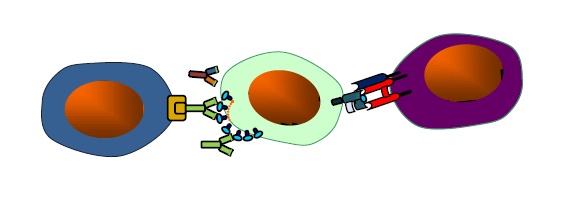
The overall goal of the laboratory is to understand the ontogeny of HIV-1 specific MHC class I-restricted and non-restricted immune responses that work by eliminating HIV-1 infected cells and how these can be induced by AIDS vaccine candidates. The studies gravitate around class I-mediated cytotoxic CD8+ T cell responses, antibody-dependent cellular cytotoxicity (ADCC), gene expression in effector cellular subsets, and development of Ab-based molecules that can engage cytotoxic effector subsets.
- Pre-clinical evaluation of Dual-Affinity Re-Targeting molecules to eradicate HIV-1 latently infected cells.
- Understanding differences between human and non-human primate effector functions of Fc-gamma Receptor-bearing cells.
- Immune monitoring of Fc-mediated Ab cytotoxic responses in pre- and clinical passive and active immunization studies.
Selected Achievements
The laboratory team has worked since 2001 on testing samples from vaccine recipients for cytotoxic T lymphocyte (CTL) and antibody-dependent cellular cytotoxic (ADCC) responses as part of the AIDS Vaccine Evaluating Group (AVEG) and, subsequently, for the HIV vaccine trial network (HVTN). The laboratory team was the first to characterize vaccine-induced cross-clade clade CD8 CTL responses and the difference in class I-restricted epitope recognition between HIV-1 infected individuals and vaccine recipients. The laboratory team followed up this initial epitope mapping of cellular responses with the epitope mapping of ADCC responses.
In 2011, the team reported the anti-C1 epitope as the most recognized epitope by ADCC ab responses in infected individuals. The laboratory has gained a unique expertise in exploring the breadth and mapping of HIV-1 epitopes recognized by the cellular and humoral responses of the immune system. Its expertise has provided essential in designing the novel class of therapeutics named DARTs in collaboration with our colleagues at UNC-Chapel Hill and MacroGenics.
Contact Us
For pre- and clinical trial studies: Dr. Guido Ferrari gflmp@dm.duke.edu or Mrs. Sherry Stanfield-Oakely: sherry.oakley@dm.duke.edu
For basic science studies: Dr. Justin Pollara: justin.pollara@dm.duke.edu
For flow cytometry questions: Mrs. Joy A Pickeral, joy.pickeral@dm.duke.edu
For ELISPOT questions: Mr. Mark Berrong, mark.berrong@dm.duke.edu
For administrative questions: Mrs. Mary Oris, mary.oris@dm.duke.edu or Mrs. Melissa Kerkau, Melissa.kerkau@dm.duke.edu
Publications and Funded Projects
View Dr. Ferrari's profile to see his publications and funded projects.
Lab Members

- Guido Ferrari, M.D., Associate Professor, PI, Director
- Justin Pollara, PhD, Assistant Professor, Co-PI, Co-Director
- Sherry Stanfield-Oakley, BS, ADCC Laboratory Manager
- Joy Pickeral, BS, Analyst II
- Mark Berrong, BS, Analyst II
- Jeremy Fitzpatrick, BS, Analyst I
- Whitney Edwards, BS, Analyst I
- Charles Beck, BS, Analyst I
- Melissa Zinter, BS, Technician II
- Kelsey Hall, BS, Technician II
- Tori Huffman, BS, Technician II
- Katelyn Faircloth, BS, Technician II
- Melissa Kerkau, MS, Program Manager